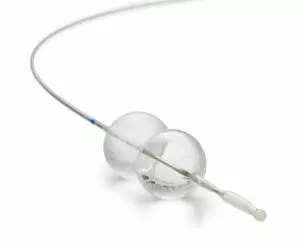
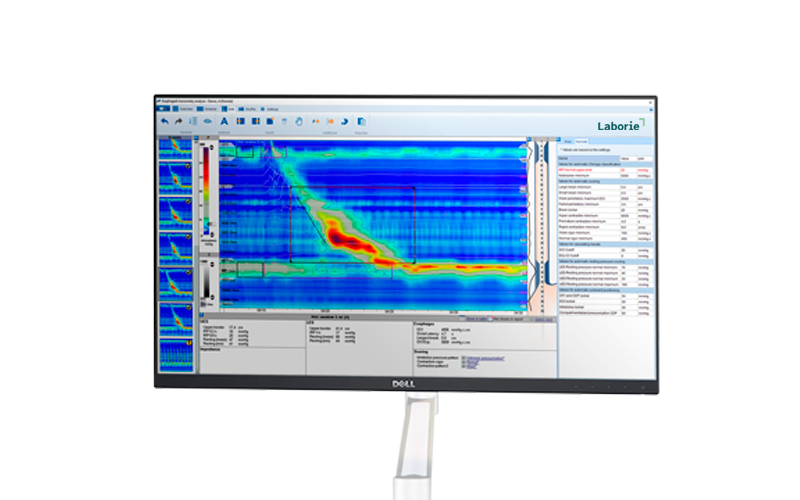
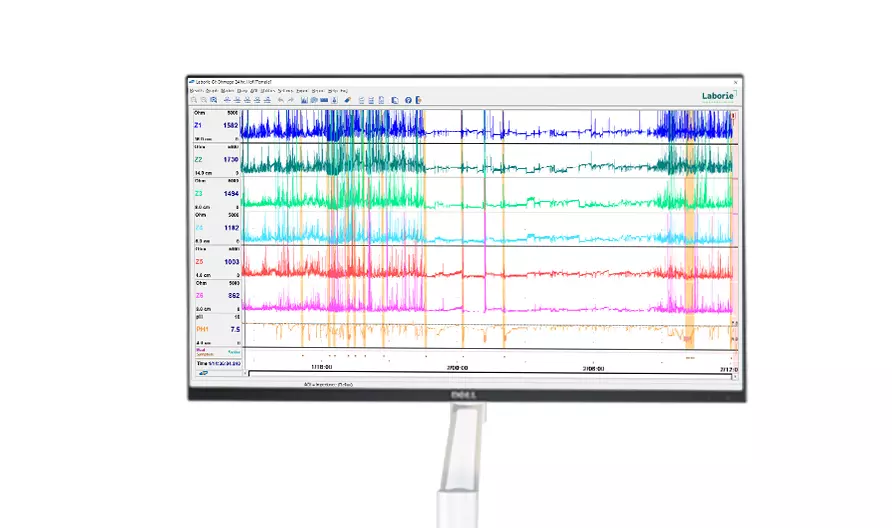
Latest Product Information
Educational Resources
Dynamic healthcare environments require that healthcare professionals stay current with the latest clinical procedures and technologies to deliver consistent clinical excellence, productivity, and a positive patient experience. Working closely with clinicians, universities and non-profits, our educational resources help you grow your knowledge. View our comprehensive list of educational resources below.
Trending News

Want To See How Laborie Can Suit Your Needs?
At Laborie, we believe that great healthcare is an essential safeguard of human dignity. Our Mission every day is to operate as a world-class specialist medical company making and advancing technologies that preserve and restore human dignity. We do that today by helping people with pelvic and gastrointestinal conditions live normal lives, and by helping mothers and babies have safe deliveries.